This is the first FDA-approved, treatment device for ADHD that can be used at home while the child sleeps. 
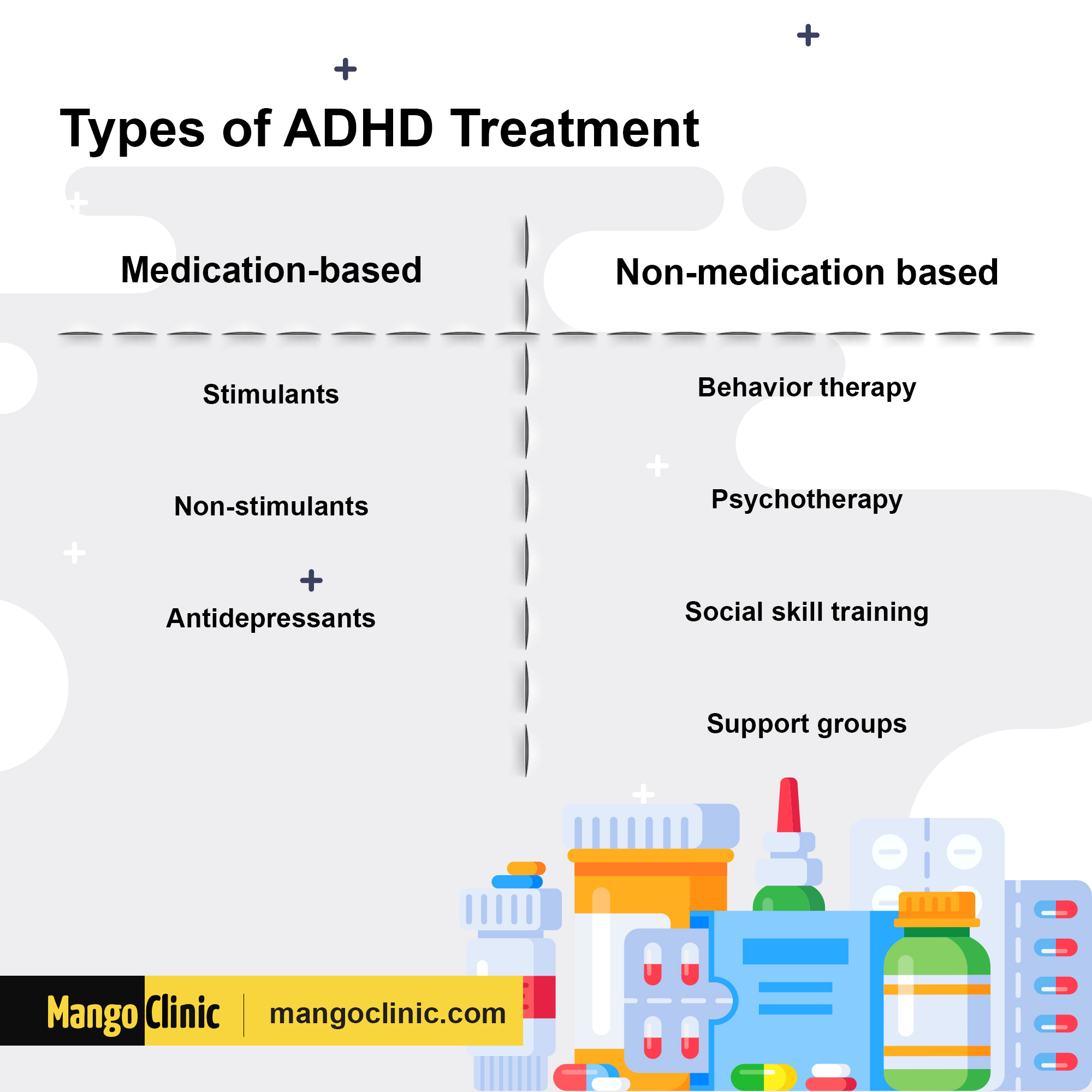
How does the device differ from other treatments? Hopefully, future research will help us identify individual characteristics that predict which treatment will likely work for that person. And there are children who don’t respond well, or at all, to stimulants, or who experience unwanted side effects. For now, the decision is made according to the clinical expertise of the treating professional in conjunction with the wishes of the parent.īut there are a good number of parents who would like to avoid pharmacological treatment, especially after a child is newly diagnosed. How do you determine which children should be treated with trigeminal nerve stimulation?Ĭurrently, there is no empirical way to predict whether a child will respond better to one treatment as opposed to another. Loo discussed the device and its potential in ADHD treatment. Ian Cook, professor emeritus of psychiatry and bioengineering at UCLA, is a co-inventor of trigeminal nerve stimulation and an author of the study he also is senior vice president and chief medical officer at NeuroSigma. James McGough, co-director of the ADHD Clinic at the Stewart and Lynda Resnick Neuropsychiatric Hospital at UCLA. The study’s co-principal investigators were Sandra Loo, a professor-in-residence of psychiatry and biobehavioral sciences at the David Geffen School of Medicine at UCLA and a member of the Jane and Terry Semel Institute for Neuroscience and Human Behavior and Dr. The device, which is about the size of a mobile phone, is loosely attached to the child’s pajamas, and it has two wires that connect to an adhesive patch that is worn on the forehead during sleep. Called Monarch eTNS, it treats the symptoms of ADHD by sending gentle electrical pulses to the trigeminal nerve, which transports sensation from the face and head to the brain, and indirectly leads to stimulation of multiple brain regions. The device tested in the study was developed using technology invented at UCLA and licensed by NeuroSigma, a Los Angeles-based company focused on developing electronic medical devices. The FDA decision followed the completion of a UCLA study demonstrating the effectiveness and safety of a trigeminal nerve stimulation device that could be a new option for treating the disorder. Stimulants, such as Adderall and Ritalin, are the most commonly prescribed ADHD treatments - and are considered to be the most effective.īut in April, the FDA approved the first device-based, non-drug therapy to treat ADHD in children. Deciding how to treat attention deficit hyperactivity disorder in children can be a difficult decision for parents. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
